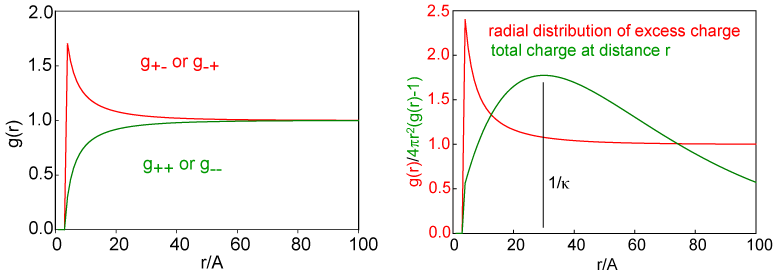
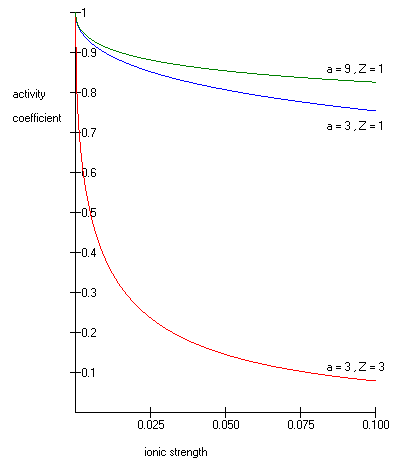
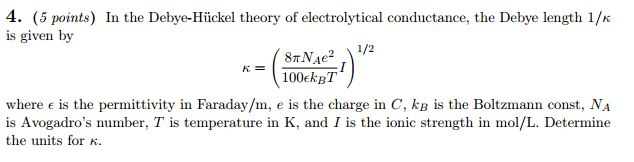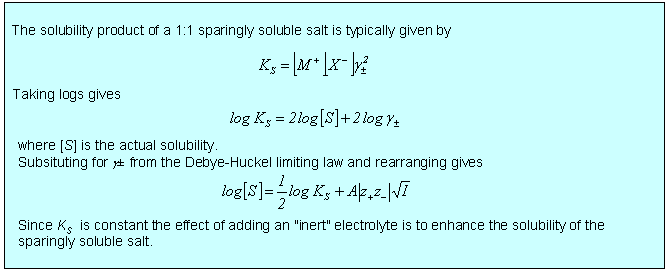The theory of electrolytes. I. Freezing point depression and related phenomena' (Debye & Hückel, 1923)

Rigorous treatment of pairwise and many-body electrostatic interactions among dielectric spheres at the Debye–Hückel level | The European Physical Journal E
The theory of electrolytes. I. Freezing point depression and related phenomena' (Debye & Hückel, 1923)

Insight into Debye Hückel length (κ−1): smart gravimetric and swelling techniques reveals discrepancy of diffuse double layer theory at high ionic concentrations | Journal of Petroleum Exploration and Production Technology

Insight into Debye Hückel length (κ−1): smart gravimetric and swelling techniques reveals discrepancy of diffuse double layer theory at high ionic concentrations | Journal of Petroleum Exploration and Production Technology

SOLVED: For an electrolyte solution, state whether the thickness of the ionic atmosphere (1/κ) (also known as Debye-Hückel screening length) increases, decreases, or remains the same when the following parameters vary. Explain
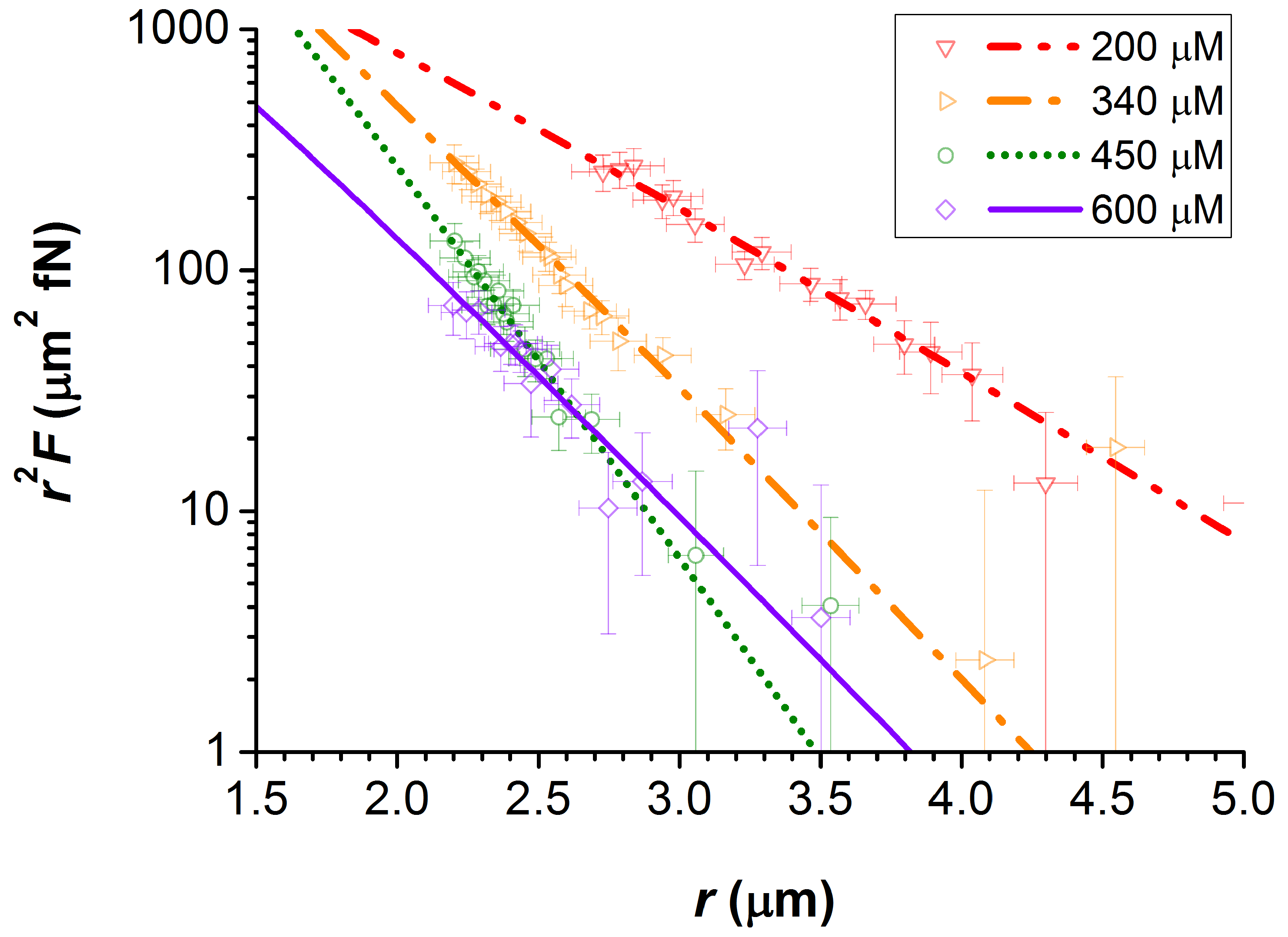
Colloids and Interfaces | Free Full-Text | Failure of Debye-Hückel Screening in Low-Charge Colloidal Suspensions

Extended Pitzer–Debye–Hückel Model for Long-Range Interactions in Ionic Liquids | Journal of Chemical & Engineering Data